Sulfuric Acid Plant

Sulfuric Acid Plant
Sulfuric Acid
Sulfuric acid is used in a wide range of processes, it is used in metal treatment, for drug manufacture, for paint, for making fertilizer and the making of explosives.
Sulfuric acid is one of the important products in the basic chemical industry, it not only as raw material for many chemical products, sulfuric acid used in the metallurgical industry and in the production of metals, particularly nonferrous metals; in the production process of petroleum products, such as gasoline and lubricating oil, concentrated sulfuric acid refining is required; and it’s also widely used in the production of fertilizers such as ammonium sulfate and calcium super phosphate; used in the production of pesticides such as copper sulfate, zinc sulfate can be used as fungicides, thallium sulfate can be used as rodenticide, ferrous sulfate, copper sulfate can be used as herbicide and so on.
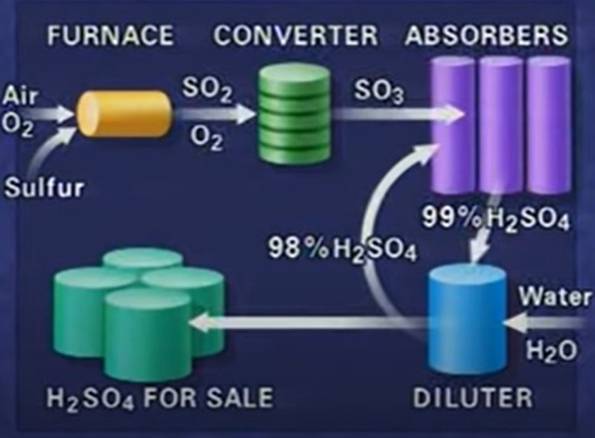
In Sulfuric Acid plant, Sulfuric acid is formed by burning sulfur in a furnace to form sulfur dioxide. In a converter the sulfur dioxide reacts with more air to form sulfur trioxide. In absorbers, the sulfur trioxide mixes with fairly concentrated sulfuric acid to increase the concentration of the acid. The acid then diluted to the concentration needed by the customer. Some acid is recycled through the absorbers. Some is stored for sale.
Sulfur Furnace
The sulfur is burnt in a furnace. Sulfur dioxide passed to the converter. Sulfur trioxide is fed to the absorption towers to produce acid of the required concentration. This is then stored for delivery by tanker.
Raw material
The Sulfur can be powder or blocks or granule or liquid. Air the second raw material is drawn from the atmosphere, dried and compressed.
Burning Sulfur
The Sulfur is sprayed into a furnace, where it burns in the dry air. Sulfur combines with oxygen in the air to form sulfur dioxide.
![]()
The oxidation produces large amounts of heat. These heat is used to boil water, to produce steam, to generate electricity. The gas mixed which leaves the furnace contains sulfur dioxide and enough oxygen for the next stage.
Catalytic Conversion
In the converter, sulfur dioxide reacts with oxygen to produce sulfur trioxide.
2SO2+O2=2SO3
The reaction requires a catalyst mainly a vanadium oxide. It is most effective at 440 degrees Celsius.

There are four beds of catalyst in the converter. The first catalyst bed converters about 63% of the sulfur dioxide to sulfur trioxide, and heat is produced. The gas stream is cooled before it passes to the next catalyst bed. Hot gas super heats steam from the furnace boiler. The gas stream passes through two further catalyst beds and the percentage conversion increases. Between each stage the hot gases have to be cooled. By the third bed, about 95% of the original sulfur dioxide.
Absorption
After the third catalyst bed, the gas stream is led into the bottom of an absorption tower packed with ceramic material. The rising gas meets a stream of hot sulfuric acid that runs down the absorber. The sulfur trioxide reacts with the water in the acid to increase the concentration of the acid to about 99%.
SO3+H2O=H2SO4
The gas from the top of the tower, still contains some sulfur dioxide and oxygen, and is fed back into the final bed of the converter after which 99.5% of the original sulfur dioxide has changesd to sulfur trioxide. The 99% concentrated acid is diluted with water to about 98%, for example, and this increases the volume of the acid. This extra 1% or so is the volume of the acid that has been made. The acid concentration is checked by sampling.
Beside sulfuric acid, the plant generates a large amount of heat which is used to produce high temperature steam and make electricity- electricity is sold to the national grid.
The production process is controlled and monitored from a control room. Acid is transported to customers by rail and by road.
Service
We provide capacity of Sulfuric Acid plant from 50TPD to 2000TPD and provide perfect construction drawing and sulfuric acid equipment; professional installation and commission on site; at the same time, we also provide the design plan according to the customer’s actual situation; and we also supply maintain service according to customers’ requirement.
Stage of Sulfuric Acid plant
 |  |  |
Belt for raw material | Burning Sulfur | Dry absorption |
 |  |  |
Dry absorption | Converter | Sulfuric acid storage tank |
Application